Lupus Responders
1. SRI Responder
Although the BILAG Index is the recognised gold standard for monitoring Systemic Lupus Erythematosus (SLE) disease activity, and SLEDAI (Selena or 2K) provides simpler, though less sensitive alternative, these indices provide a measure of disease activity in a patient between visits. Due to the multisystemic nature and complexity of lupus, there was no standardized method for defining response to therapy, or comparing complex activity manifestations across a large cohort of patients. Growing insight into the pathogenesis of inflammatory diseases like SLE has led to the introduction of specific targeted biologic therapies. To investigate the efficacy of these new biologic agents, disease activity must be monitored regularly by a reliable and validated instrument. Recent studies on new biologics for treatment of SLE have started to use a new composite measurement for disease activity and response in SLE. This new response to disease activity measure is known as the SLE Responder Index (SRI), and utilises criteria from three different internationally validated indices, the British Isles Lupus Assessment Group (BILAG 2004), SLEDAI 2K (or SELENA-SLEDAI), Physician Global Assessment (PGA).
SRI response is defined as
1) a >= 4-point reduction in SELENA–SLEDAI score,
2) no new BILAG A or no more than 1 new BILAG B domain score, and
3) no deterioration from baseline in the physician’s global assessment by >= 0.3 points.
2. BICLA Responder
An alternative rresponder to the SRI is the BILAG-based Composite Lupus Assessment (BICLA), which is also a composite index that was originally derived by expert consensus of disease activity indices . It was employed as the primary endpoint in the EMBLEM trial of epratuzumab in lupus (moderate-to-severe SLE disease activity), where it appeared to discriminate well between standard of care and epratuzumab added to standard of care, at least in some doses tested.
BICLA response is defined as :
(1) at least one gradation of improvement in baseline BILAG scores in all body systems with A or B disease activity at baseline
(2) no new BILAG A or more than one new BILAG B scores;
(3) no worsening of total SLEDAI score from baseline;
(4) no significant deterioration (≤10%) in physicians global assessment and
(5) no treatment failure (initiation of non-protocol treatment).
Wallace D, Strand V, Furie R, Petri M, Kalunian K, Pike M, et al. Evaluation of treatment success in systemic lupus erythematosus clinical trials: development of the British Isles Lupus Assessment Group-based composite lupus assessment endpoint. Arthritis Rheum. 2011;63:S894
SRI v BICLA Responder
A key difference to acknowledge between SRI and BICLA outcomes is that BICLA requires only partial improvement but in all organs, whereas SRI will respond to full improvement in some manifestations, and not necessarily in all organs. Accordingly SRI is more likely to indicate a targeted system response in patients with multiple systems involved at baseline. BICLA will indicate overall patient improvement. Thus either instrument could be used an optimal primary endpoint depending on the population under study and the design of a given clinical trial.
BLIPS Responders
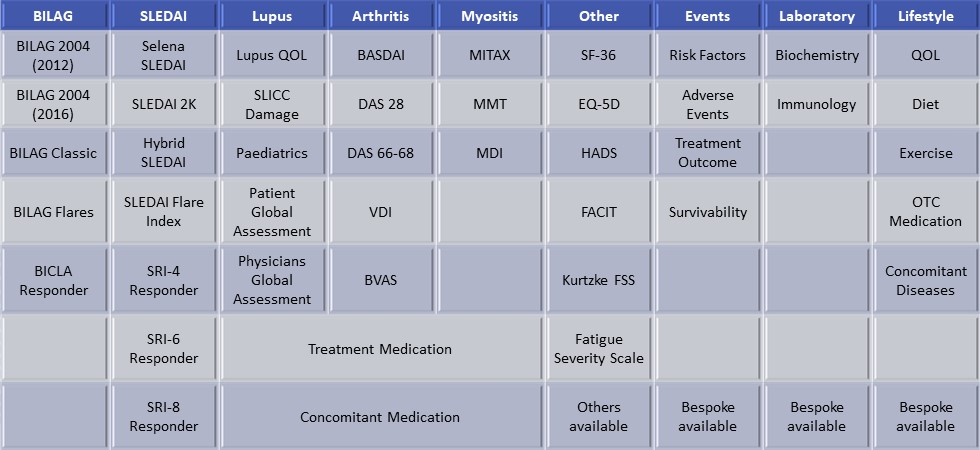
BLIPS is already validated to calculate A scores, B scores, Flares and SLEDAI totals, hence it is the ideal tool to automatically determine SRI and BICLA Responders. The traditional method of calculating responders, is to compare baseline with the final visit in a study. However this does not give the complete picture. As shown in a patient example below, there is the posibility that the patient can have a flare which is not picked up by a single, end of study, SRI measurement. The BLIPS Responder™ module will illustrate whether the patient is responding at every visit. This has been succesfully combined with BLIPS Alert™ to automatically alert physicians, in studies where the protocol demands a reduction in (e.g) corticosteroid treatment due to a level of response.



 1
1
 2
2
 3
3
 4
4
 5
5
 6
6
 7
7
 8
8
 9
9
 10
10